Research library
Longer briefings when you want depth after a guide.
The Peptide Renaissance
You and Peptides Research · ~10 min read
A strategic briefing on therapeutic peptides
Where the science came from, what is working, what is risky, and how regulators are responding. Covers the GLP-1 boom, the SNAC oral-delivery breakthrough, the regenerative gray market, the 2023 FDA Category 2 ban, and the AI-driven discovery frontier.
Peptide Mechanisms — Deep Dive
You and Peptides Research · ~12 min read
The detailed mechanics behind the briefing
A longer companion that goes deeper on the SNAC mechanism step-by-step, the AI / computational design toolkit, market projections through 2033, and the real-world dosing constraints behind oral peptide therapies.
How Peptides Are Made — A Manufacturing Deep Dive
You and Peptides Research · ~11 min read
From Merrifield’s 1963 resin bead to flow chemistry, recombinant DNA, and modern hybrid plants
The most important technology behind every approved peptide drug is not a receptor or a delivery system — it is the chemistry that lets you build the molecule in the first place. This is how peptides are actually manufactured today: SPPS, microwave SPPS, flow chemistry, recombinant biology, and the hybrid plants that combine all of them.
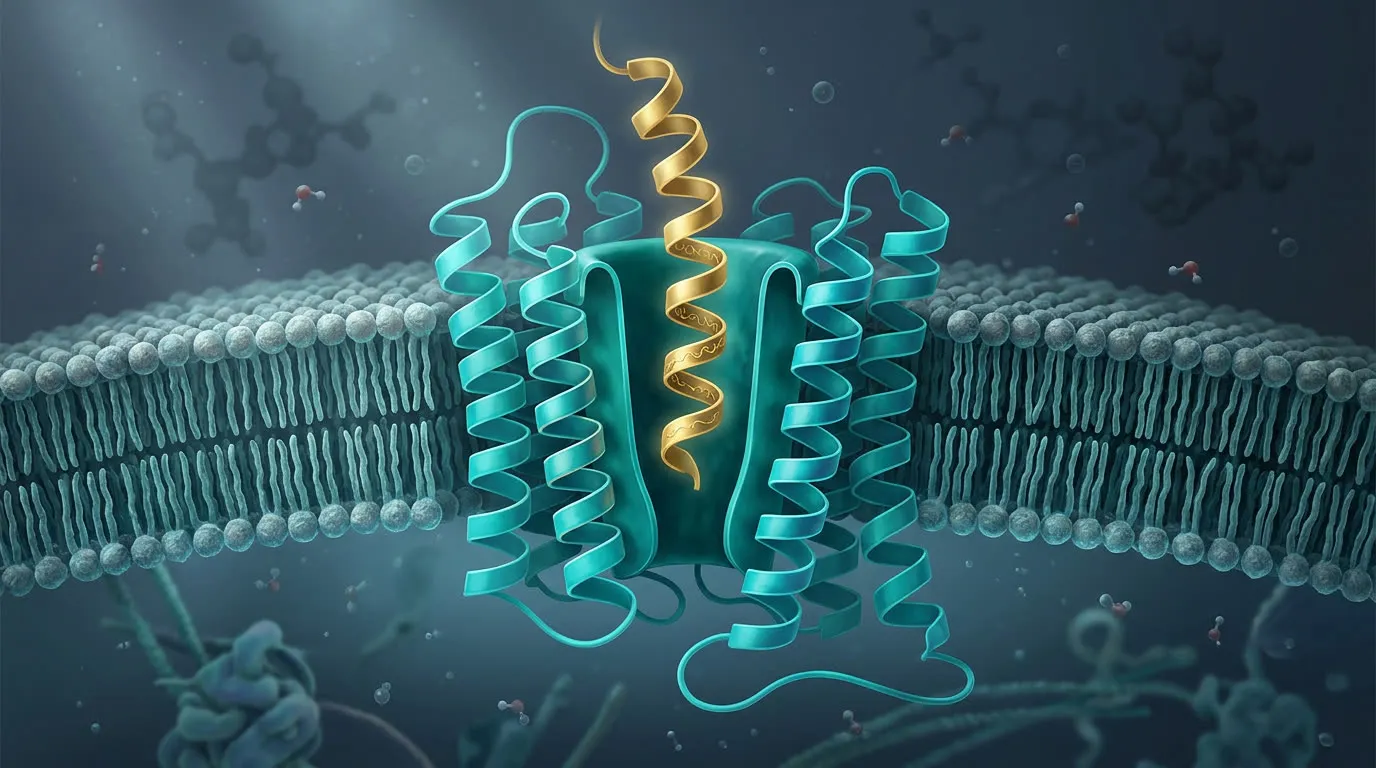
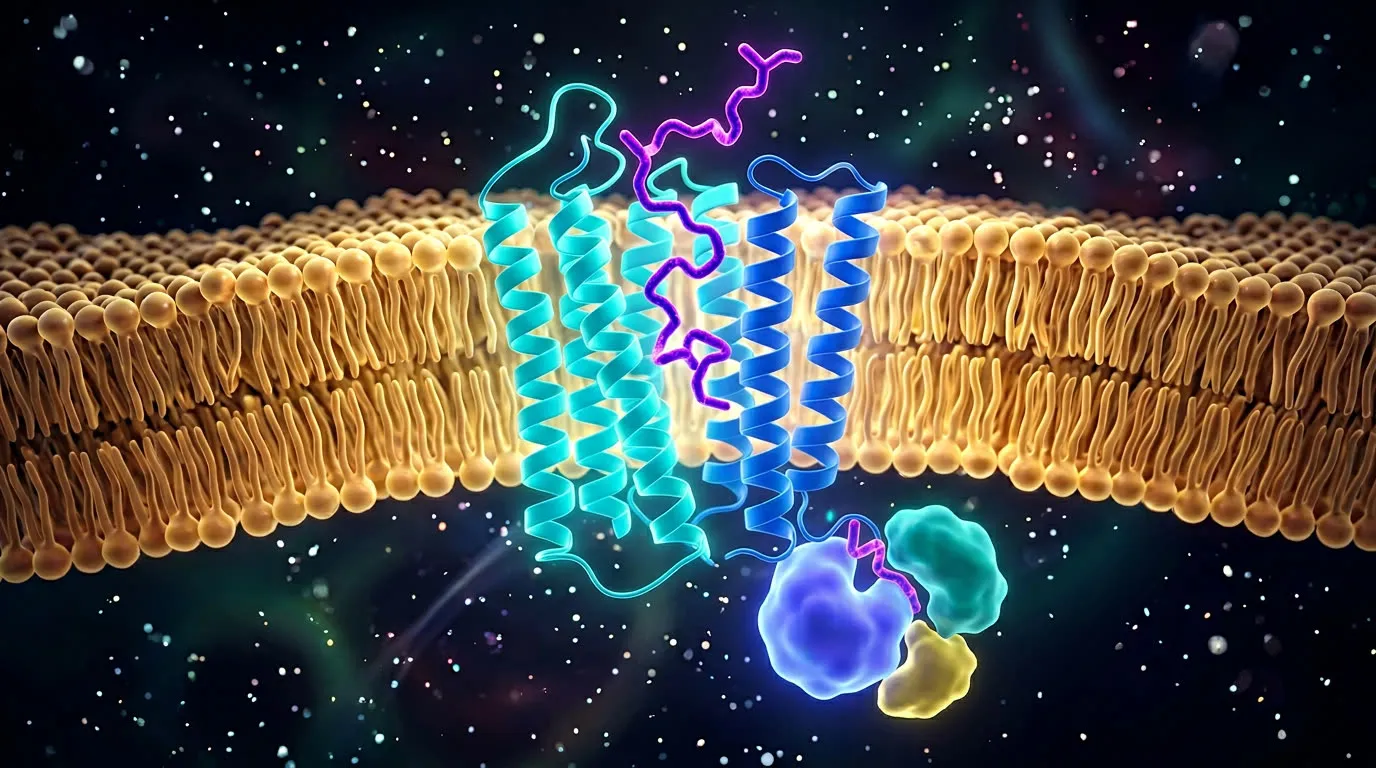
How Cells Read the Telegram
You and Peptides Research · ~10 min read
Receptor-ligand binding, GPCR cascades, and the picomolar problem
Peptides are signaling molecules — chemical telegrams that bind receptors on the surface of cells and trigger downstream effects. This briefing walks through the kinetics of receptor binding, the GPCR family that handles most peptide drugs, what "picomolar" actually means, and why some peptides survive in the bloodstream for an hour while others survive for a week.
The 2026–2027 Compounding Question
You and Peptides Research · ~9 min read
What the FDA Pharmacy Compounding Advisory Committee’s next review actually changes
In 2023 the FDA placed 19 peptides — including BPC-157, CJC-1295, and Ipamorelin — on Category 2, blocking compounding pharmacies from making them. The Pharmacy Compounding Advisory Committee (PCAC) revisits the 503A Bulks List in 2026–2027. This is what is actually being decided, what each plausible outcome looks like, and what it means for the field.